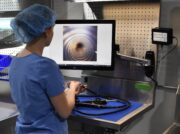
What is a borescope, exactly? 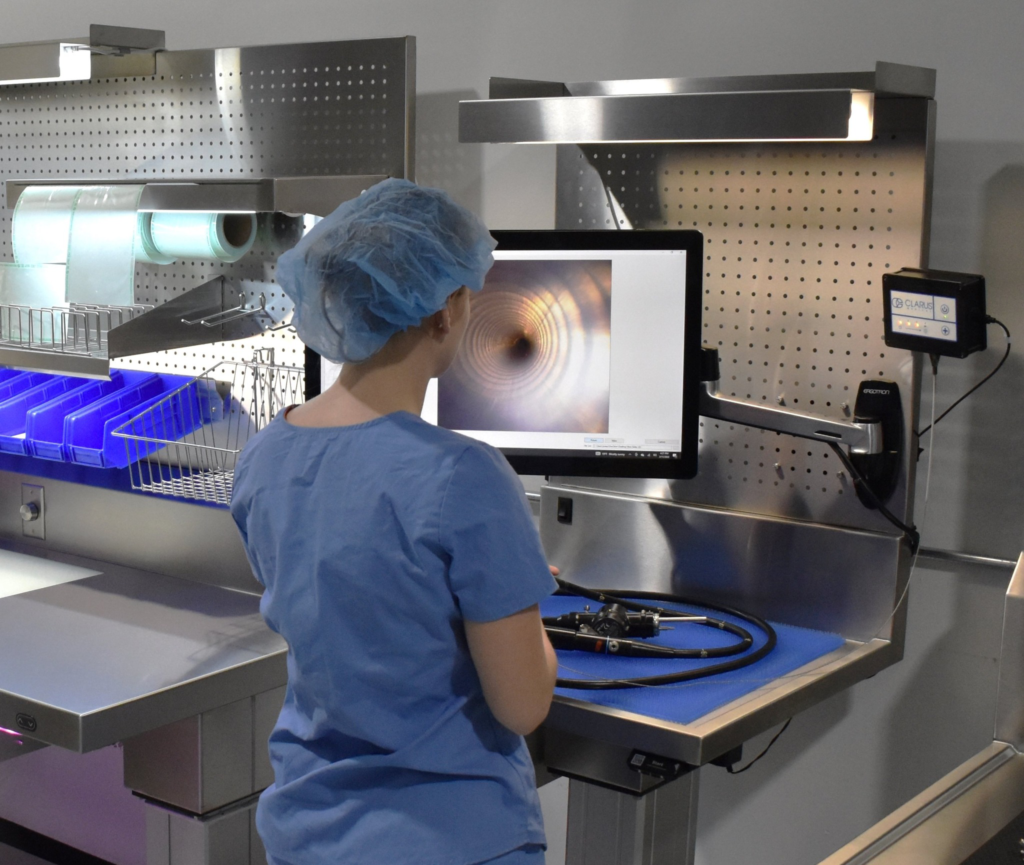
A borescope is a flexible inspection device with a camera and light that allows technicians to see inside the internal channels of an endoscope or lumened instrument. Essentially, a scope for a scope!
In sterile processing departments and GI labs, borescopes are used to inspect:
- Internal channels of flexible endoscopes
- Elevator channels in duodenoscopes
- Lumens of surgical instruments
- Internal surfaces that cannot be seen externally
A borescope provides real-time visual confirmation of cleanliness and device integrity.
Why are borescopes becoming more widely used?
Even when instruments appear clean, functional or correct on the outside, internal channels can be hiding moisture, damage, bioburden or other variables which impact high-level disinfection or sterility. Borescopes provide a more 360-view of instrument cleanliness.
What specific problems do borescopes detect?
A borescope can detect many issues, including:
- Residual bioburden or ‘leftovers’ from cleaning, such as brush bristles
- Moisture retention
- Internal scratches
- Channel damage
- Peeling materials
- Cracks or deterioration
When should borescopes be used?
Borescopes are typically used after manual cleaning and before high-level disinfection (HLD) or sterilization. Common inspection points include:
- After manual cleaning
- During quality assurance checks
- After repairs
- During routine inspection programs
- When contamination is suspected
This ensures cleaning was effective before the endoscope is reused.
How do borescopes improve patient safety?
Borescopes are one of the truest ‘safety stops’ in an instruments reprocessing cycle. Before mistakes can go further down the chain, there is a chance to correct improper practices.
HLD or sterilization cannot happen if instrumentation isn’t clean. Patients can be injured when our instruments aren’t functional.
What instruments should be visually inspected with borescopes?
Borescope inspection is particularly useful for:
- Shavers
- Flexible endoscopes
- Complex components, such as elevator channels (borescopes give magnified views of anything and can be valuable even if not being thread into a channel).
Borescopes are particularly important with high-risk endoscopes, such as duodenoscopes, colonoscopes or gastroscopes. These endoscopes frequently have elevator channels, moving distal tips, and narrow internal lumens.
What are the benefits of borescopes for the GI or SPD department themselves?
It’s a huge win for patient safety when borescopes are implemented into workflows. But for the GI lab or sterile processing, borescopes can also deliver value internally by:
- Identifying poor equipment; residual brush bristles typically mean low-quality brushes, or improper brushing technique
- Improved accountability with instrument repair services
- Stronger internal reputation for the department, when instrument repair dips overall, and quality outcomes improve.
What are some of the challenges with using borescopes?
Borescopes can present a couple key challenges:
- Damage to the borescope itself; borescopes are often incredibly delicate, and may be expensive to repair.
- More questions than answers: just because something was found inside an instrument channel doesn’t clarify how to reconcile the issue.
- Borescopes come with technological requirements: ensuring staff are trained on computer use, computers are available and functional and kept up to date
- Borescopes inspection requires space; ensure your department has adequate space & electrical to set-up borescopes stations
What should be done when something is found inside an instrument?
First, a department policy should be written for your unique situations. However, general recommendations include:
- Reprocess the instrument again; any visual bioburden or debris should attempt to be removed
- Take photos or videos and seek second guidance on if the finding may be damage
- Log & track identified issues, and create a library of ‘next steps’ for technicians or nurses to reference, and track overall patterns.
Should our department have a borescopes program?
This answer depends entirely on your department’s objectives, resources and instrument inventory. However, departments that meet the following should strongly consider a borescope program:
- Any flexible endoscopes are being reprocessing, especially high-risk endoscopes
- Your goal is to verify cleaning effectiveness, not just work off assumptions
- Infection prevention initiatives are a top priority
- You adhere to ANSI/AAMI ST91, SGNA or AORN guidelines
- Your department has actively struggled with instrument repair costs, visual debris during cleaning, high endoscope volume, or failed cleaning audits
Borescopes matter when it comes to quality assurance programs, and the output of our reprocessing cycles. While borescopes are not the comprehensive answer to effective QA outcomes, they are a key element to a more 360-view into the effectiveness of our efforts. Borescopes can be a powerful tool in the quality assurance toolkit.
To learn more about quality assurance programs, visit our CE-accredited Reprocessing Report, The Age of Quality Assurance: A Complete Guide to Quality Assurance in Endoscope Reprocessing.
To get started on evaluating borescopes for your facility, start with our PureClear™ Inspection Scopes.