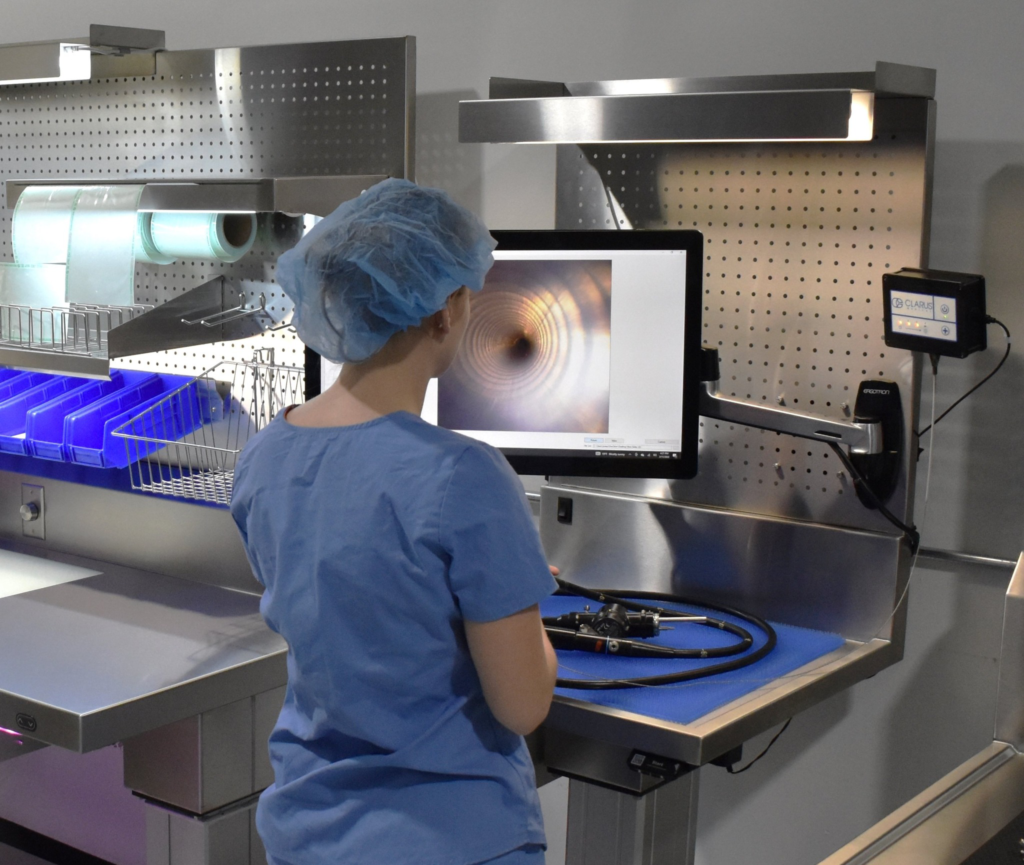
Borescopes have become a staple component of comprehensive quality management systems, and rightfully so. They provide insight into the unseen, verifying the effectiveness of cleaning practices through visual 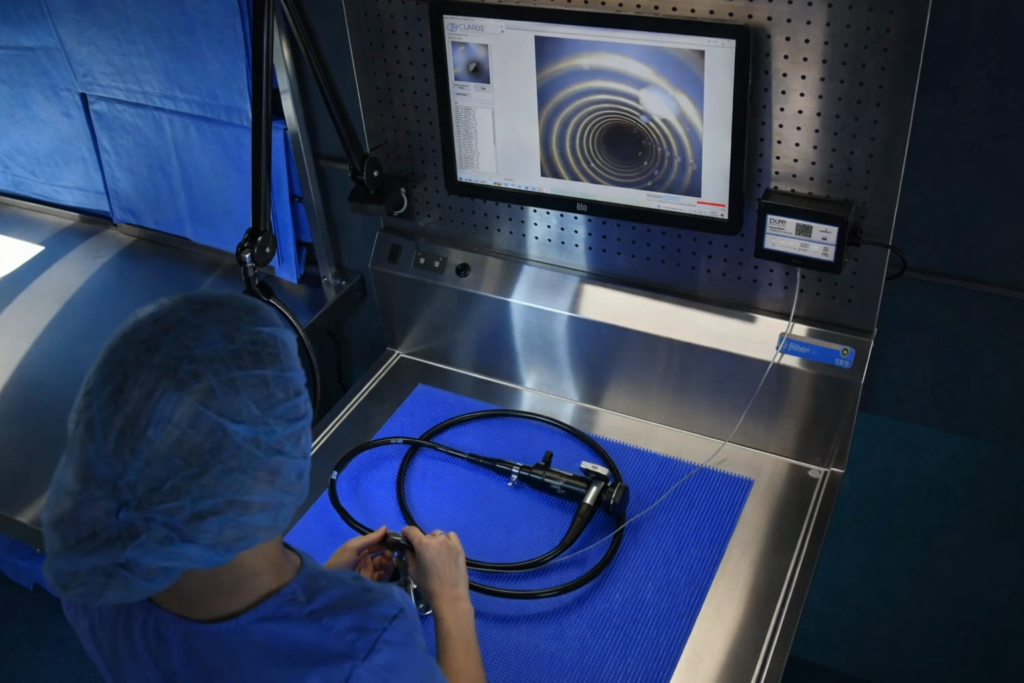 inspection. With AI capabilities, some models can even analyze the condition of channels and identify residual soil within the inspected lumen.
inspection. With AI capabilities, some models can even analyze the condition of channels and identify residual soil within the inspected lumen.
Beyond identifying pass-or-fail quality outcomes, borescopes allow teams to detect and categorize findings such as retained bioburden and instrument damage. Identifying these discrepancies early helps prevent patient contamination, additional device damage, and potential infection transmission.
The standards provide clear guidance and rationale for incorporating borescopes into our processes and quality management systems.
Elevated Practices
Enhancing visual inspection is just one benefit of implementing borescope practices into reprocessing workflows. Borescopes are designed to verify process effectiveness. While they do not directly prevent damage or improve cleaning processes, the findings gathered through their use directly influence necessary improvements.
Borescopes substantiate findings and provide visual evidence of process quality output, as well as instrument condition. This supports early identification of discrepancies and can help prevent additional damage before it progresses further in the cycle.
As stated in ANSI/AAMI ST79 Annex D:
“A number of methods can be used to evaluate the results of the cleaning process. The most common method is a visual inspection, sometimes involving the use of a lighted magnifying glass for inspecting cleanliness of device surfaces or the use of borescope cameras for inspecting the internal channels of lumened instruments.”
Inspection as a Critical Step
When ANSI/AAMI ST91 was published in 2022, it identified six (6) scopes categorized as high risk (ANSI/AAMI ST91 Section 7.8.4). These scopes are known for complex designs that create cleaning challenges and increase patient safety risks.
The scopes include:
- Duodenoscopes
- Linear Ultrasound (EUS) scopes
- Bronchoscopes
- Endobronchial ultrasound (EBUS) scopes
- Ureteroscopes
- Cystoscopes
- Others as determined by the facility
These scopes carry a “shall” statement focused on cleaning verification. A “shall” requirement gives reprocessing teams a clear mandate to verify cleaning effectiveness and catch failures before patient use. Cleaning verification is now required for these high-risk scopes after every use.
But what does that look like in practice?
ANSI/AAMI ST91 Annex F defines the components of a cleaning verification process:
“Ideally, cleaning verification should include:
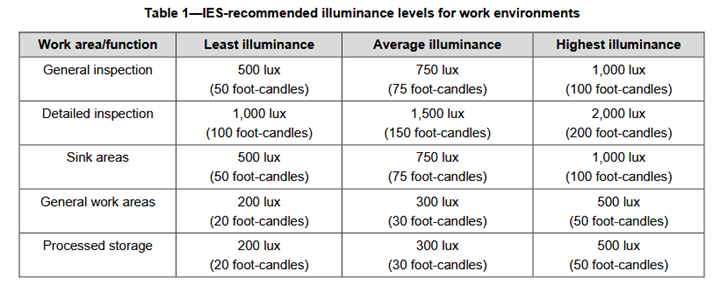
a) visual inspection combined with other verification methods that allow the assessment of both external surfaces and the inner housing and channels of medical devices;
b) testing the cleaning efficacy of equipment and the cleaning process; and
c) monitoring key cleaning parameters (e.g., temperature). Manufacturers provide users with such tests so that medical devices can be tested directly after cleaning in a way that will not damage the device or require recleaning.
A more objective and sensitive method than visual inspection is to measure the levels of organic soil on the cleaned device. There are commercially available tests that allow users to rapidly verify that adequate cleaning has been performed.”
Cleaning verification incorporates two (2) types of inspection:
- Visual inspection (e.g., use of a borescope for internal components)
- Cleaning efficacy testing that measures residual bioburden
Both methods complement one another and provide reprocessing teams with critical information. This data supports risk assessments and analytics that either verify process efficiency or indicate that adjustments are necessary.
With data collected from cleaning verification, we can track patterns and trends and drill down to specifics, allowing for precision decision-making. Whether it is a specific scope that fails consistently or a time of day when discrepancies increase, targeted improvements become possible.
Maybe the data shows a need to increase scope inventory. Maybe it justifies adding staff to support throughput demands. When supported by objective evidence, those initiatives carry significantly more weight in strategic planning.
Cleaning verification supports patient safety while strengthening process reliability for our teams.
How to Implement
Adding a new process to the workflow can feel daunting and may be met with scrutiny, particularly because it naturally adds time. When implementing a value-added process, preparation is critical.
First, clearly define the issue you are looking to solve. It may be as simple as aligning with current standard expectations. In other cases, you may be addressing a known performance gap. Conduct a risk assessment and determine how the process adds measurable value — this forms the foundation for implementation.
Second, evaluate available product features and capabilities in the market. Identify which solutions align with your department’s goals and operational expectations.
Third, determine where the new process fits within your workflow. How will it impact ancillary steps? Is there a strategic location that makes the most sense? Department layout and manufacturer IFUs will often influence inspection points and workflow design.
Finally, processes are only as strong as the training behind them. Provide structured education, opportunities for hands-on practice, and space for questions. Equip staff with the why, the how, and the expectations. The more prepared teams are before implementation, the more confident they will be in both the process and its intended outcomes.
Borescopes are a major value-add to quality management systems in both GI and SPD reprocessing departments. With thoughtful implementation, they can reveal critical insights into cleaning processes and the factors that influence efficiency.
Curious to learn more about borescopes? Check out our FAQ blog on borescope here: https://pure-processing.com/blog/borescopes-101-faq-for-sterile-processing-and-gi-lab-professionals/
References:
ANSI/AAMI ST79: 2017


 are positioned as helpful, available when needed most, and designed to set hospitals up for success. However, the introduction of loaner trays into workflows can result in serious consequences in both process and outcomes. It’s important to be aware of the dangers when bringing loaner trays into SPD. The consequences of poorly implemented loaner programs are far-reaching.
are positioned as helpful, available when needed most, and designed to set hospitals up for success. However, the introduction of loaner trays into workflows can result in serious consequences in both process and outcomes. It’s important to be aware of the dangers when bringing loaner trays into SPD. The consequences of poorly implemented loaner programs are far-reaching.


 obstacles include:
obstacles include:
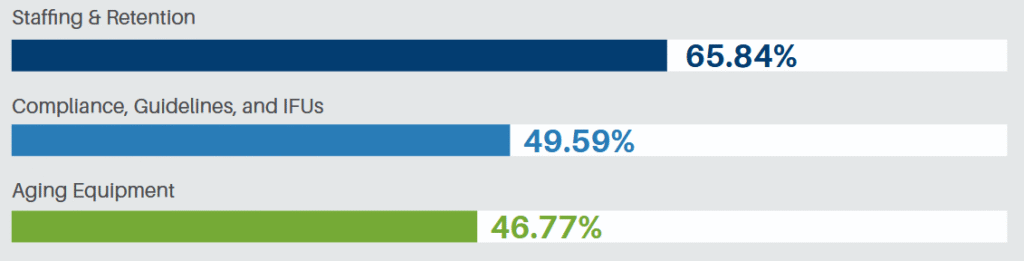
 processing leaders are applying AI today. The themes were pragmatic. Teams want tools that support standards, training, and staffing without disrupting existing workflows.
processing leaders are applying AI today. The themes were pragmatic. Teams want tools that support standards, training, and staffing without disrupting existing workflows. processing leaders are applying AI today. The themes were pragmatic. Teams want tools that support standards, training, and staffing without disrupting existing workflows. Below are three use cases you can try without overhauling your systems or processes.
processing leaders are applying AI today. The themes were pragmatic. Teams want tools that support standards, training, and staffing without disrupting existing workflows. Below are three use cases you can try without overhauling your systems or processes.