Workflows can be found in any industry. The Cambridge Dictionary defines workflow as “the way that a particular type of work is organized, or the order of the stages in a particular work process.”
This step-by-step progression is what creates processes and generates outcomes. There are many factors that impact the output from a workflow & its processes. It is critical to patient safety and staff success that each factor be considered during a workflow evaluation and planning phase to ensure desired outcomes are achieved and potential challenges are mitigated.
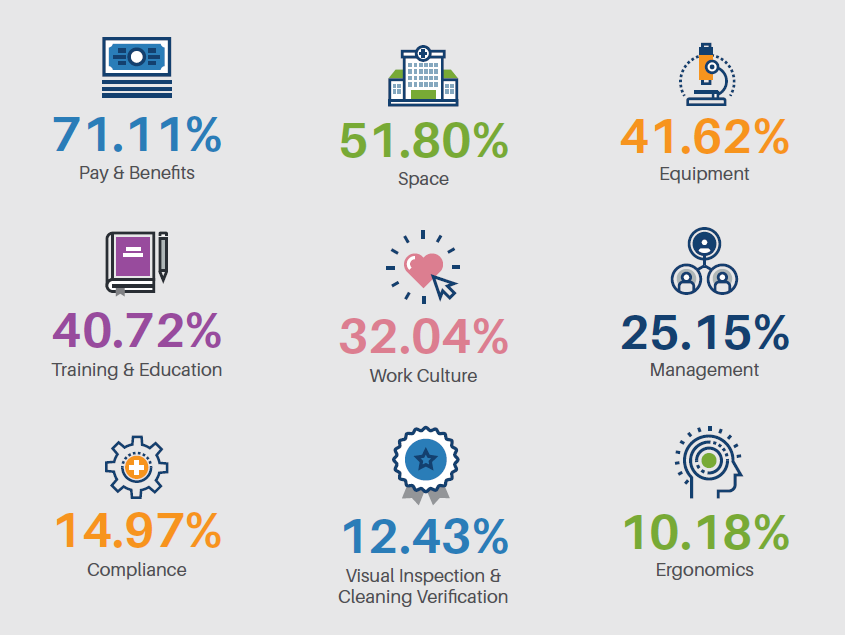
Limited Space
Real estate in sterile processing and GI departments can be lacking and improvisions can occur that will lead to missteps or risk of contamination at certain points of a process. However, limited space can also impact overall safety outcomes for both staff and patients.
ANSI/AAMI ST79 Section 3.3.6 speaks to the importance of planning out your space and equipment based on the volume of work to be done there:
Ergonomic factors affecting worker safety and comfort should be considered when designing workspaces in the decontamination area, including…
b) adequate space to maneuver, queue, and unload carts or other transportation means at times of average daily peak workload;
Without the space required to queue up and maneuver around staged case carts or instruments on the assembly side can lead to frustration or injury.
Clutter, Overcrowded Workspaces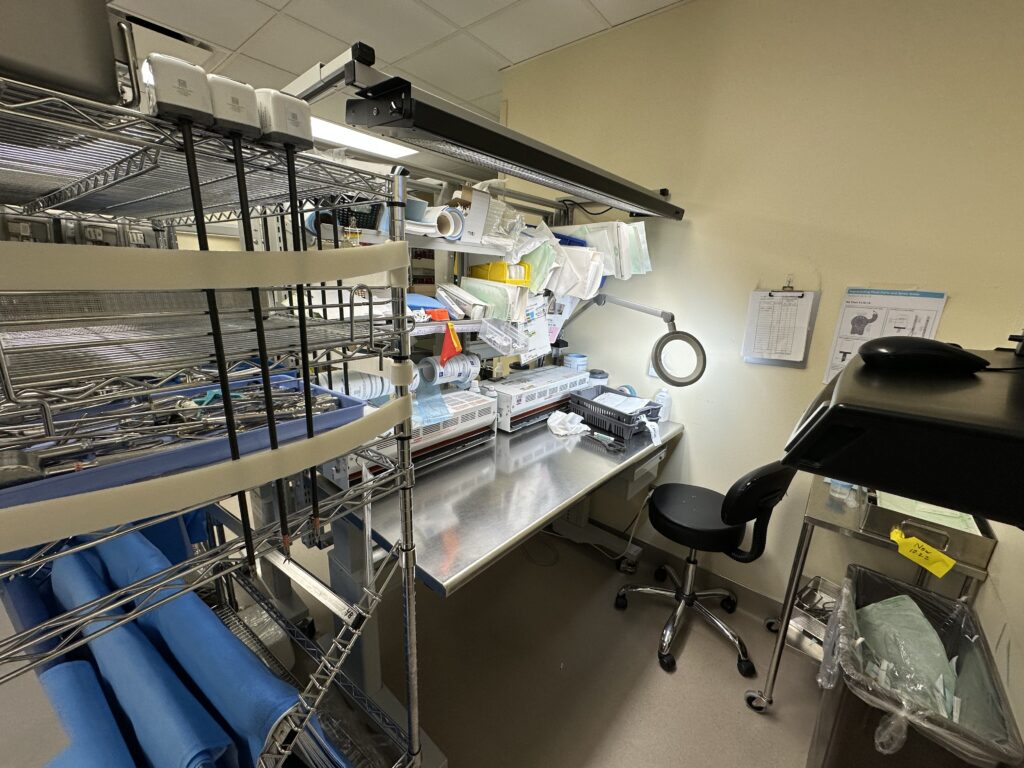
Clutter and overcrowded workspaces can also take up mental space. Frustration, distraction and delays can all be attributed to the condition of the spaces in our workflows. Cluttered workspaces lead to discrepancies, gaps and overall non-compliance. Some studies have also been cited to show that clutter or overcrowded spaces can lead to higher stress levels. (IH, 2018)
Often, workstations are shared spaces, which can mean that each person will interact with the workspace slightly differently than the last. Without a reset of the work area, clutter will continue to accumulate.
Additionally, clutter can develop over time as new technologies and resources get added to a work area. A constant state of adding without subtracting what is no longer relevant can also lead to overcrowded workstations. This also leads to safety risks including fire hazards, dust collection and mold formation. (IH, 2018)
As things collect and pile up, real estate decreases while stress and safety risks increase.
Poor Organization
Organization can encourage compliance and even guide a workflow. Conversely, disorganization can have the opposite effect. Disorganization lends to confusion, misplaced steps and unclear expectations. Rather than focusing on the task at hand, the mind and eyes wander to other tasks or distracting messes that can lead to longer productivity times, lower throughput and diminished employee morale.
Staff who spend as much time cleaning up, reorganizing or making sense of disorganized stations in a workflow increase the risk of making mistakes or losing inventory and tools.
Siloed Workflows
Carol Cain, in the handbook, Patient Safety and Quality, discusses the pitfalls of viewing workflows as separate processes:
“Most often, when workflow processes are looked at in isolation, the processes appear quite logical (and even efficient) in acting to accomplish the end goal. It is in the interaction among the processes that complexities arise. Some of these interactions hide conflicts inthe priorities of different roles in an organization, for example, what the nursing team is accountable to versus the physician team and its schedule. Organizations also adapt workflows to suit the evolving environment. Over time, reflecting on organizational workflows may show that some processes are no longer necessary, or can be updated and optimized.” (NIH, 2008)
When we don’t consider what is happening before or after a specific portion of the process, we lose vital information that can impact how a workflow is maintained. Interdepartmental partnerships are another great example of workflow processes that influence outside of specific sterile processing or GI practices. Transitional workflows, such as instrumentation moving from decontamination to assembly, also influence outcomes and efficiency as instruments progress through the process. Evaluating where workflows overlap can break down these silos and improve the outputs from both workflows.
Workflow development takes the study of time, environment, space and outcomes fully understand what the impact is going to be on the department and the intended outcomes.
Interested in learning more about workflow and processes? Check out these additional blogs to take a deeper look!
Thief of Time: Workflow Optimization’s Role in Time Management
Don’t Let the Work Around Become the Workflow – Optimizing Your Department with Data
References
Cain C, Haque S. Organizational Workflow and Its Impact on Work Quality. In: Hughes RG, editor. Patient Safety and Quality: An Evidence-Based Handbook for Nurses. Rockville (MD): Agency for Healthcare Research and Quality (US); 2008 Apr. Chapter 31. Available from: https://www.ncbi.nlm.nih.gov/books/NBK2638/ https://dictionary.cambridge.org/us/dictionary/english/workflow
ANSI/AAMI ST79:2017 https://www.utphysicians.com/the-stress-of-mess-how-clutter-can-affect-your-workflow/ https://intermountainhealthcare.org/blogs/can-organizing-impact-your-mental-health



 so intricately connected to another that missing one can cause workflows and processes to falter.
so intricately connected to another that missing one can cause workflows and processes to falter.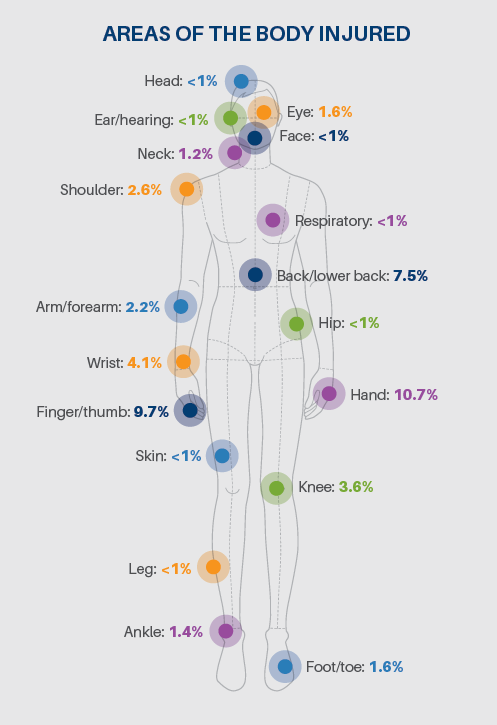
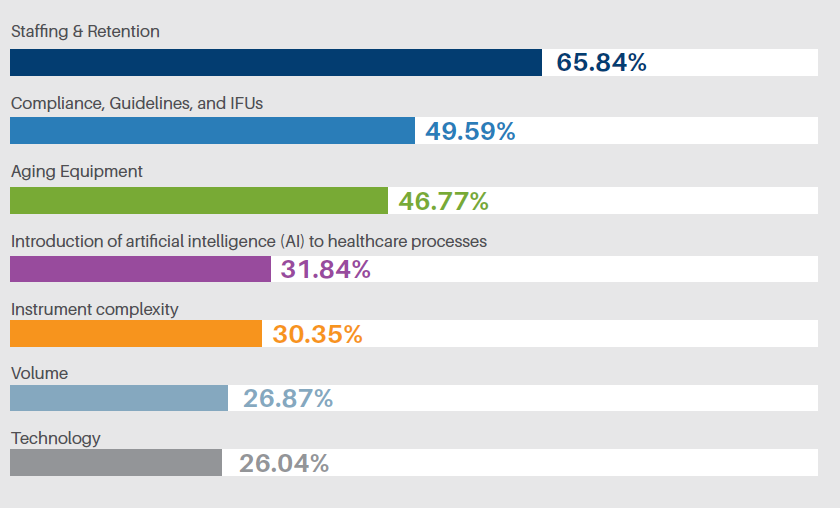
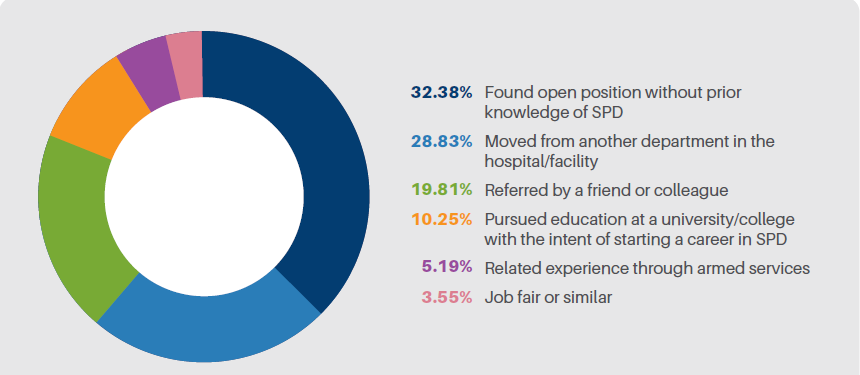
 across healthcare, from clinical standards to health system enterprises. The intent behind standardization is to improve quality, optimize available resources and reduce preventable errors. However, are there situations where maybe the opposite can occur in standardization? When standardization overlooks nuances resulting in errors, inconsistencies and glossing over specific steps? Standardization can then have a negative effect on our goals.
across healthcare, from clinical standards to health system enterprises. The intent behind standardization is to improve quality, optimize available resources and reduce preventable errors. However, are there situations where maybe the opposite can occur in standardization? When standardization overlooks nuances resulting in errors, inconsistencies and glossing over specific steps? Standardization can then have a negative effect on our goals.




