One of the most important places in an endoscopy suite or clinic is the decontamination sink. Undervaluing the impact that a well-designed endoscopy reprocessing sink has jeopardizes all proceeding steps in the process. The sink is the center for leak testing, manual cleaning, and visual inspection procedures. A poorly designed or suited endoscopy sink can increase cross-contamination risks, damage delicate endoscope inventory, cause bottlenecks, or create confusion and cause lapses in reprocessing.
Standards have clarified what elements are required for a properly designed endoscopy sink. What are some of these design elements, and how do they positively impact endoscope reprocessing outcomes?

Separation & Workflow
Separation between clean and dirty has long been a challenge for endoscopy reprocessing departments because of space. Space is a non-negotiable factor for creating separated clean versus dirty areas. Space also impacts workflow; without it, nurses and technicians double-back on each other, potentially cross-contaminating cases and scopes.
When one-way workflow cannot be enforced in an endoscope processing room, high-volume and backlogs create confusion. It’s easy to lose sight of proper workflow when scopes are piling up and the clock is ticking.
Sink design can have a positive impact. A properly designed endoscopy sink should consider the full decontamination space. Is my sink installed where doors don’t impede one-way workflow? If there’s only one room, can I maintain at least 4 foot of space between clean and dirty (ANSI/AAMIST 91:2021)?
Limited space does not have to limit solutions. If space is of concern, how can my sink vendor provide partitions and design elements that reinforce proper workflow, and separation between clean dirty? These are valid considerations for sink design.
| “Adequate space shall be provided to allow for the manual cleaning and rinsing of devices during decontamination. It is optimal that the manual cleaning area is physically separated by walls or partitions in a two-room configuration to control contaminants generated during manual cleaning. Doors that open in the direction of the one-way workflow and closeable pass-through windows separating the decontamination area from the adjoining disinfection/sterilization area should remain closed when not in active use. An endoscopy processing room with a one-room design should provide a minimum of 4 feet between the decontamination area and the clean work area and either a separating wall or a barrier that extends a minimum of 4 feet above the sink rim to separate soiled work areas from clean work areas (FGI, 2018 [146]; AORN, 2018e [39]).”
ANSI/AAMI ST91 4.2.2 Physical Separation |
Basins
Most flexible endoscopes have at least two characteristics in common: they are delicate, and intricately designed. These qualities demand careful handling and specialized care during cleaning.
While basins should be deep enough to allow for full submersion of the endoscope, excessively deep basins can lead to ergonomic challenges for staff, risking discomfort and potential injury from prolonged bending. However, striking the right balance is important, as appropriately deep basins help to mitigate aerosols during cleaning.
Basins should also be a certain size: over-coiling is a common risk for larger scopes. Over-coiling also reduces leak testing outcomes, masking potential leaks.
“Use decontamination sinks that are deep enough to allow complete submersion of the endoscope and large enough to allow the endoscope to be positioned in the sink without tight coiling”
AORN Guidelines for Perioperative Practice 1.9.1
| “Use decontamination sinks that are deep enough to allow complete submersion of the endoscope and large enough to allow the endoscope to be positioned in the sink without tight coiling”
AORN Guidelines for Perioperative Practice 1.9.1 |
Having at least 2 sink basins, optimally 3, prevents cross-contamination between scopes. Sinks should also be height-adjustable, when available, to reduce ergonomic discomfort for staff. Improper ergonomics can have severe, negative consequences for worker safety and retention. In the 2024 GI Nurse and Technician State of the Industry report, 189 anonymous endoscopy participants ranked the ergonomics in their department at a 6.3/10 score. This is a current challenge, and can ‘leak’ into the cleaning process through mistakes, and shortcuts.
| “Sinks should be deep enough to allow complete immersion of the endoscope to minimize aerosolization. The size of the sink should be adequate (i.e., a minimum of 16 inches x 30 inches) to ensure that the endoscope can be positioned without tight coiling. Sinks should be height-adjustable so that personnel do not have to bend over to clean endoscopes. An ideal decontamination sink is height-adjustable, approximately 36 inches (91 centimeters [cm]) from the floor and 8 to 10 inches (20 to 25 cm) deep, enabling a person of average size to work comfortably without undue strain on the back; foot stools should be readily available to accommodate shorter personnel.
At a minimum, two sinks or one sink with two separate basins should be used. Consideration should be given for additional space for delayed processing protocol. One sink or sink basin should be designated for leak testing and manual cleaning and the other only for rinsing. Optimally, three sinks or one sink with three separate basins should be used, with each function in a separate sink or basin.” ANSI/AAMI ST91 4.3.2 Sinks and accessories |
Staging Space
The staging space serves multiple crucial functions in endoscopy processing areas. Firstly, it ensures that each endoscope is treated as an individual case, preventing any risk of cross-contamination or damage that may occur from stacking them. This is essential for maintaining the integrity of the endoscope and the safety of patients.
Secondly, the staging area provides an optimal environment for inspection and leak testing, allowing technicians to thoroughly examine each endoscope for any potential issues before use.
Additionally, the staging area can facilitate visual inspection procedures, enabling staff to assess the cleanliness and condition of the endoscope before proceeding with processing.
Reprocessing professionals and their vendor partners should consider where and how staging space is integrated into the sink design.
| Sinks should have attached solid counters or adjacent work surfaces on which to place the endoscope while attaching it to the leak tester, detach and separate removable components, and to inspect the endoscope for physical damage and cleaning effectiveness.”
ANSI/AAMI ST91 4.3.2 Sinks and accessories |
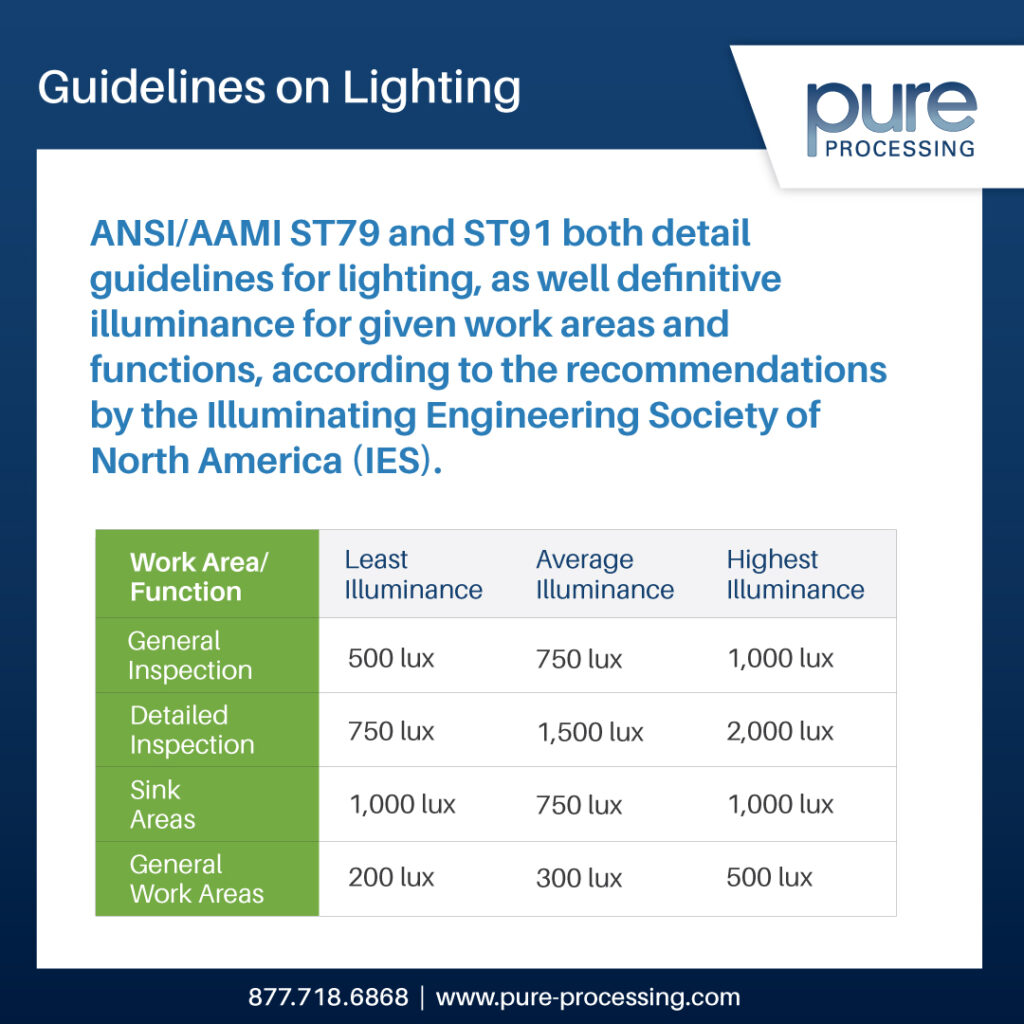
Lighting
Fine detailed cleaning is routine with endoscopes. Per ANSI/AAMI ST91: 2021, wet leak testing should now be conducted for 60 seconds to help find fine bubbles, tricky in poor lighting conditions. Multiple ports and channels mean more areas for cleaning, and more areas to miss. Lighting is critical to ensure proper visual inspection procedures. Magnifiers further adjunct lighting for proper inspection.
| “Lighting of the recommended illuminance should be placed above the sink and counter area so that personnel can adequately perform inspection activities as the endoscope is processed (see 4.3.8 and Table 1). A lighted magnifier should be available for inspection.”
ANSI/AAMI ST91 4.3.2 Sinks and accessories |
Gastroenterology clinics and sterile processing departments struggle with many challenges, most of which aren’t going away. A practical solution to these challenges can be equipment design, and understanding why standards make certain recommendations.
Endoscopy and sterile processing professionals should carefully consider which vendors they partner with to fulfill these design needs. Pure Processing intimately understands and studies endoscope reprocessing standards and departments, to help provide custom endoscope sink solutions. Contact us to learn more about our process, and if we’re the right fit for your project!
Looking for a checklist to start your endoscope sink evaluation? Download our free, Endoscope Reprocessing Checklist, and ensure your next GI sink meets these 16 key considerations!



 departments: educators. They play a pivotal role in reprocessing departments, but not all departments have, or are able to have, a dedicated educator role.
departments: educators. They play a pivotal role in reprocessing departments, but not all departments have, or are able to have, a dedicated educator role.