Our Voice of the Customer (VOC) council convened earlier this spring to discuss a topic on the minds of many sterile processing department managers, but not necessarily at the forefront of current  conversations: data.
conversations: data.
During our panel discussion, a few bullet points were recognized as important by nearly all committee members:
- How to gather data
- Creating processes using that data
- Solving problems with data
- Creating departmental scorecards
While new, Reprocessing Report blog posts regarding each of these topics are upcoming, here are some high-level takeaways from the VOC call:
How to gather data
The first step towards using data in sterile processing departments is ensuring that there is a means to collect it. Our VOC members each had different ways of collecting quality data in their departments, and identified a few ways to improve data collection:
- Make a commitment to collecting data at the department level
- Utilize more features of tracking systems that may already be in place
- Increase communication between sterile processing department and the operating room
Keep an eye out for our upcoming blog post, “Studying Your Department: How to Gather Data” to learn more.
Creating processes using data
A key advantage around data was the ability to use it as a base to create functional, efficient processes. For example, a reduction in output because of a standard or guideline change may require additional headcount or technology investments. In order to maintain compliance and output levels, data can help sell these investments internally.
Data can also be used to remedy processes by offering an objective assessment of performance. Our VOC council noted that in many departments the “workaround becomes the workflow”; what was once a temporary solution to a problem quickly become the standard. Analyzing processes using data can enable sterile processing and gastroenterology managers to identify workarounds that have taken root, and take actions to correct them.
Departmental problem solving
Data can be used to identify and solve a myriad of problems in sterile processing departments. Our VOC council explored how data can solve problems in their departments:
- Use key leading indicators (KLI’s) to identify bottlenecks and productivity concerns to address issues proactively
- Create audit programs to gauge and address quality issues
- Tie performance to volume
- Gain insights into the root cause of problems; data can help give context and identify underlying problems causing surface-level issues
Creating departmental scorecards
With all this discussion about data, it was only fitting that the conversation concluded with creating scorecards that departments can use to organize and track critical data. Some important considerations discussed, included:
- Identifying what information is most important to your department
- Ensuring the accuracy of the data collected
- Finding the right solution to build, store and manage your scorecard
- Determining capabilities, features, and limitations of existing tracking software
- Identifying the resources and people you may need to ensure ongoing, accurate data collection.
These are just some of the high-level takeaways from our Voice of the Customer council meeting. Stay tuned for deep-dives into each of these data discussion points in upcoming Reprocessing Reports, and help your department begin leveraging data to benefit your department!


 adjustable sinks to accommodate employees of different heights and reduce the risk of workplace injuries. However, the budget of many departments don’t often allow for immediate upgrades to height-adjusted sinks. In those cases, a more affordable and practical solution can be a is a
adjustable sinks to accommodate employees of different heights and reduce the risk of workplace injuries. However, the budget of many departments don’t often allow for immediate upgrades to height-adjusted sinks. In those cases, a more affordable and practical solution can be a is a  such as carpal tunnel syndrome and other musculoskeletal disorders. To address this risk,
such as carpal tunnel syndrome and other musculoskeletal disorders. To address this risk,  and cleaning instruments. This requires optimal lighting that illuminates the working area and provides a clear view of the instruments. Many sterile processing and gastroenterology departments struggle with optimal lighting, which has a direct impact on eye strain.
and cleaning instruments. This requires optimal lighting that illuminates the working area and provides a clear view of the instruments. Many sterile processing and gastroenterology departments struggle with optimal lighting, which has a direct impact on eye strain. optimal height for each individual. However, with the use of
optimal height for each individual. However, with the use of 


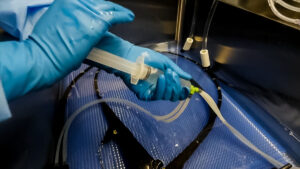 comes to no surprise, due to high turnover of both staff and scopes. With manual cleaning consuming a large chunk of time, use of automated flushing systems during the manual cleaning stage of endoscope reprocessing can result in more efficient and effective results compared to manual flushing. The devices built for automated flushing are designed to provide consistent flow and pressure during flushing. When two facilities used the Flush Buddy Systems, they found and increase in efficiency and consistent flush times.1
comes to no surprise, due to high turnover of both staff and scopes. With manual cleaning consuming a large chunk of time, use of automated flushing systems during the manual cleaning stage of endoscope reprocessing can result in more efficient and effective results compared to manual flushing. The devices built for automated flushing are designed to provide consistent flow and pressure during flushing. When two facilities used the Flush Buddy Systems, they found and increase in efficiency and consistent flush times.1  flushing with a syringe. According to
flushing with a syringe. According to 
 Many sterile processing department managers know how it feels to learn that your facility is about to undergo a Joint Commission (JC) survey; even for the most prepared departments, it can be anxiety-inducing.
Many sterile processing department managers know how it feels to learn that your facility is about to undergo a Joint Commission (JC) survey; even for the most prepared departments, it can be anxiety-inducing.
 well knowing how to readily access, retrieve, and present them, demonstrates that your department is well organized, and has done the work to ensure policies and procedures are established and accessible.
well knowing how to readily access, retrieve, and present them, demonstrates that your department is well organized, and has done the work to ensure policies and procedures are established and accessible.