
Shary Williams
Implementation of robotics programs can be problematic for sterile processing departments. The increased volume, need for additional knowledge, training requirements, and equipment can cause more than a few headaches.
We spoke with Shary Williams, Sterile Processing Quality and Education Manager at Northshore University HealthSystem to get insights on how to make a robotics implementation smooth in SPD.
Awareness
The most important factor for successful preparation in SPD ahead of a robotics program implementation is knowing that it’s happening. In many circumstances, sterile processing departments aren’t brought into the fold ahead of time, resulting in inadequate preparation and inability to properly support the new program.
Williams’ SPD Director, who had a strong relationship with the OR and was an effective communicator, made sure that each facility was in-the-know and that everyone was aware of their role in preparation.
For departments who aren’t adequately prepared for changes in workload or case volumes, mitigating the impact is the next best practical solution. Investing in back-up equipment, cross-training employees or having a general knowledge of upcoming changes and trends in instrument reprocessing can make last-minute changes less disruptive.
Training
Many technicians may be unfamiliar with robotic devices and unsure of what they are or how to properly reprocess them. “Some members of the team looked at the instruments and thought robotics would be easy!”, Williams explained.
But, as Williams continued, robotic devices are actually very complicated, and there’s a lot that a sterile processing team needs to know and understand before robotic instruments start showing up in a department.
Organizing training for your team in the weeks, and even months, before a robotics program is launched is critical to ensuring that you’re ready to go when the time comes to start supporting a robotics program.
Equipment
The preparation for a robotics program often includes the procurement of new tools and equipment to help the department reprocess robotics efficiently and in accordance with IFU. Implementing these tools, however, isn’t as simple as getting them set up in the department. Williams emphasized the need to have new tools and equipment ready one to two weeks ahead of implementation to allow technicians the time to be trained and familiarized with them.
This training shouldn’t be the sole responsibility of educators, though. Scheduling vendors to come in and conduct in-services and provide general support for new tools and equipment will enable your team to ask questions and gain a deeper understanding of how to effectively use them.
Staffing
Williams also explained that coordination between the OR and SPD was critical because staffing levels need to be increased on days when robotic surgery is planned. With extended soaking times, increased volume, and complex IFU, it’s advantageous to have more team members available to keep up, especially early on.
As a department becomes more familiar and efficient with robotic instrument reprocessing, increased staffing levels may not be as necessary.
Include the Operating Room
NorthShore’s SPD and OR teams put a tremendous emphasis on ensuring they are communicating regularly and keeping each other in the loop. A great way to get support, understanding, and collaboration from your OR counterparts? Invite them over!
As a Quality & Education Manager, Williams said that offering to include the OR team during in-services, educational events, and training helped to ensure that both teams understood the complexity of the devices and diligence required to support a robotics program.
Conclusion
Due to the team-centric, comprehensive planning of Williams and her team, the robotics program implementation went smoothly. Take a page from her playbook and remember these key factors when you’re planning to support a robotics program:
- Have effective communication to ensure that you’re aware that a robotics program is coming.
- If you struggle with a lack of communication at your facility, mitigate the impact by having back-up plans, and preparing for the unexpected.
- Train your team ahead of time; don’t wait for game day to start preparing.
- Get tools and equipment in early and lean on your vendor partners to prepare your team.
- Ensure that you have an adequate number of staff on-hand on days when robotic surgeries are taking place.
- Invite members of the OR team to attend in-services, educational opportunities, and training so that everyone’s on the same page.
Preparing for a new robotics program? Check out our Robotics Reprocessing Toolkit!



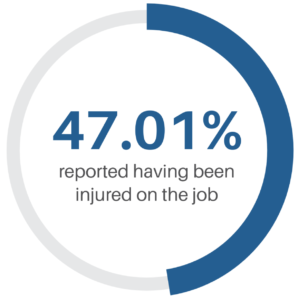

 but often gets overshadowed: Data. We partnered with Gene Ricupito, Senior Project Manager, Sterile Processing at UCSF Health to do a deep dive on solving problems using data.
but often gets overshadowed: Data. We partnered with Gene Ricupito, Senior Project Manager, Sterile Processing at UCSF Health to do a deep dive on solving problems using data. ultimately providing a “data map” of throughput in the department.
ultimately providing a “data map” of throughput in the department.









 might be valuable to have, the complexity of collecting it could make it difficult to reliably obtain. However, with a little creativity, you might be able to collect metrics B and C and get the same insights you would have gotten from A.
might be valuable to have, the complexity of collecting it could make it difficult to reliably obtain. However, with a little creativity, you might be able to collect metrics B and C and get the same insights you would have gotten from A. automated flushing systems ensure that the appropriate volume of solution is flushed per device IFUs, meaning compliance comes at a much lower cost in time spent and repetitive motion.
automated flushing systems ensure that the appropriate volume of solution is flushed per device IFUs, meaning compliance comes at a much lower cost in time spent and repetitive motion.
