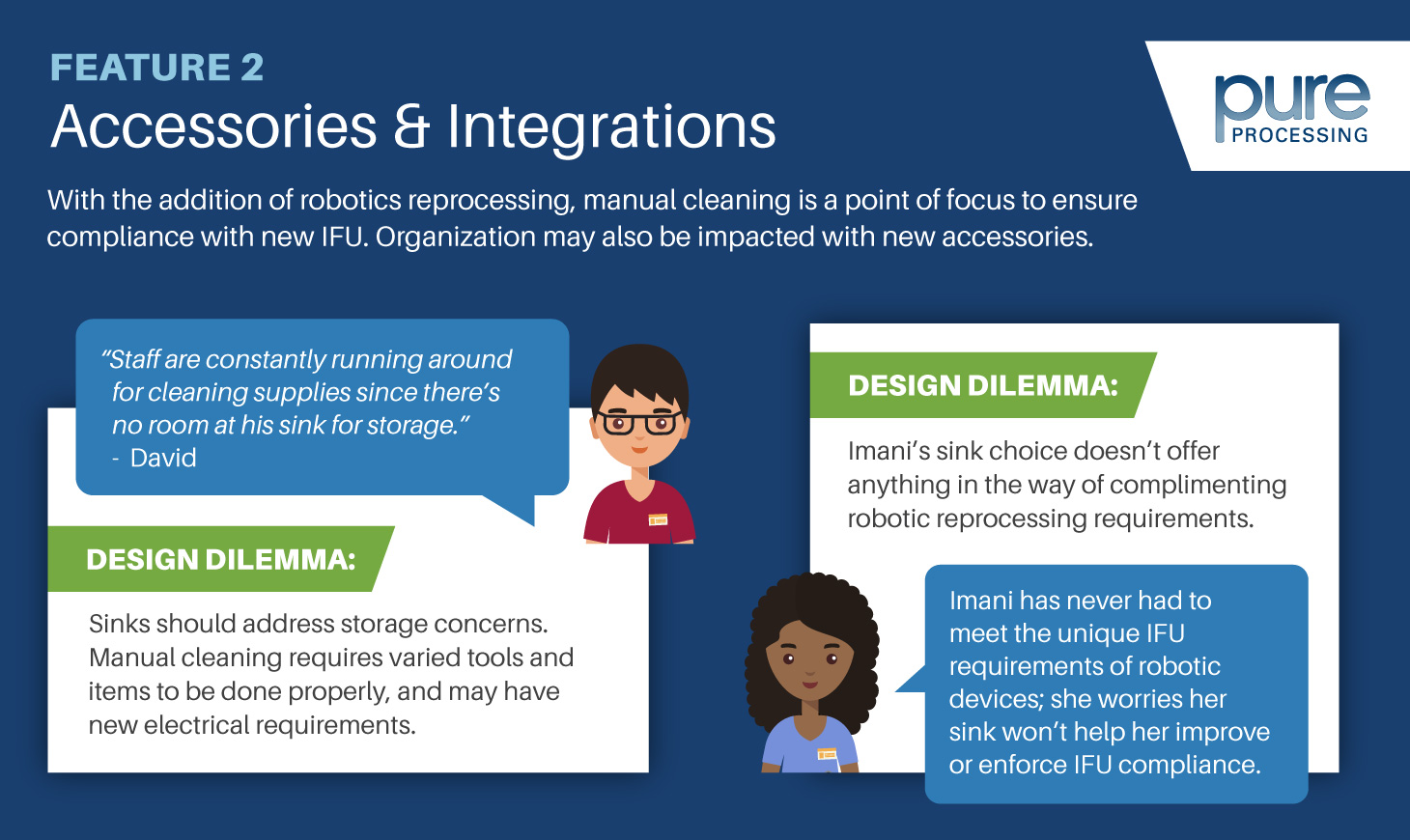
One thing most SPD professionals can relate to is a lack of space within their departments. Most central sterile departments were built into hospital plans before the innovations of the OR took place, locking departments into layouts that cannot be expanded or easily increased.
As instrument complexity and sizes grow, and as new reprocessing technologies emerge, real estate in central sterile departments will be at even more of a premium than it is today.
So, what are some strategies to get more out of limited space?
(Click the image)
Identify underutilized space
Making time to analyze and assess unused space throughout your department can illuminate many opportunities. Freeing up valuable space that’s currently utilized inefficiently, or incorporating innovative organization and storage ideas, can add efficiencies and have quite an impact.
An often low-hanging fruit is to look at the vertical space near sinks and assembly tables. While some sinks and prep and pack tables allow racked shelving to be attached to their back wall for basic storage of small materials, many departments don’t utilize this space at all. Check out our blog post on elevating your workspace for a deeper dive on vertical space usage at workstations.
Find ways to free up space
Various tools and equipment are used throughout sterile processing or an endoscopy’s reprocessing area. From computers, to borescopes, to magnifiers, there’s no shortage of tools required to help technicians or nurses do their job.
While these tools are immensely helpful and play a critical role in ensuring patient safety, they can take up a considerable amount of space. Finding alternative methods to store these tools is a worthy investment. A great example is getting computers off of surfaces by mounting above or to the side of the workstation. Some computer mounts allow for housing or handheld scanners, or laptop stands that easily fold away to reduce its overall footprint when not in use. Mounts can also help organize messy electrical and USB cords.
Areas below sinks and workstations tend to be underutilized as well. By adding open configuration shelving, you can begin storing enzymatics and chemicals that are required at that station. Integrating equipment built with this kind of storage is easier than retrofitting obsolete equipment, however.
Add functionality to existing space
Versatility is the name of the game. Being able to give space multipurpose functionality effectively doubles it. A few examples of adding functionality to an existing space include:
- Using removable staging panels over sink basins adds temporary staging space.
- Implementing prep & pack tables with built-in lights allows technicians to use the surface as both a wrapping surface as well as tool to inspect wrap with before use.
- Make deep sinks more accessible to every technician with sink inserts.
- Use small, mobile stations that can be tucked into unused corners to add dynamic soaking and rinsing capabilities or counter space wherever your department needs it without overhauling your layout.
Get a fresh perspective
Sometimes working within a space can make it difficult to see new opportunities for improvement. Allowing an outside perspective to have a look and offer suggestions can expedite the process of solving space-related issues. Like the feeling of being stumped on a puzzle and having someone else walk over and immediately place a piece, sometimes you just need a second set of eyes on a problem to identify the best solution.
Interested in exploring ways you can improve your space usage? Download our newest ‘Study Guide for Space’ checklist!






 Specifically, for lumened devices, AORN states:
Specifically, for lumened devices, AORN states:



 importantly, patient safety. All too often tools, such as magnifiers, are kept somewhere other than workstations, or deliberately moved from workstations because they inhibit technicians when not in use.
importantly, patient safety. All too often tools, such as magnifiers, are kept somewhere other than workstations, or deliberately moved from workstations because they inhibit technicians when not in use. All too often, departments centralize blue wrap where space allows. Its also common that the central location isn’t located where wrapping takes place.
All too often, departments centralize blue wrap where space allows. Its also common that the central location isn’t located where wrapping takes place. they can get in the way if they are not given a deliberate, permanent home. Assembling a rigid container becomes difficult with a keyboard in the way. Inspecting an instrument is also challenging when a monitor blocks the magnifier.
they can get in the way if they are not given a deliberate, permanent home. Assembling a rigid container becomes difficult with a keyboard in the way. Inspecting an instrument is also challenging when a monitor blocks the magnifier. Inspecting and packaging requires immense attention to detail; not only does everything need to be executed flawlessly to ensure patient safety, but the correct materials and equipment need be employed based on the types of instruments and the sterilization processes they require. A variety of small issues can arise.
Inspecting and packaging requires immense attention to detail; not only does everything need to be executed flawlessly to ensure patient safety, but the correct materials and equipment need be employed based on the types of instruments and the sterilization processes they require. A variety of small issues can arise. than just divert their focus; it can slow them down considerably. A good process produces good results, and good processes require organization. After all, it was an industrial level of organization that made Henry Ford’s production line so successful.
than just divert their focus; it can slow them down considerably. A good process produces good results, and good processes require organization. After all, it was an industrial level of organization that made Henry Ford’s production line so successful. Let us briefly revisit the meal you are preparing to impress your spouse. The first thing you would probably do is gather your ingredients. You head to the fridge for a protein and vegetables, then to the pantry for some pasta, and finally grab a few spices from the cabinet.
Let us briefly revisit the meal you are preparing to impress your spouse. The first thing you would probably do is gather your ingredients. You head to the fridge for a protein and vegetables, then to the pantry for some pasta, and finally grab a few spices from the cabinet.

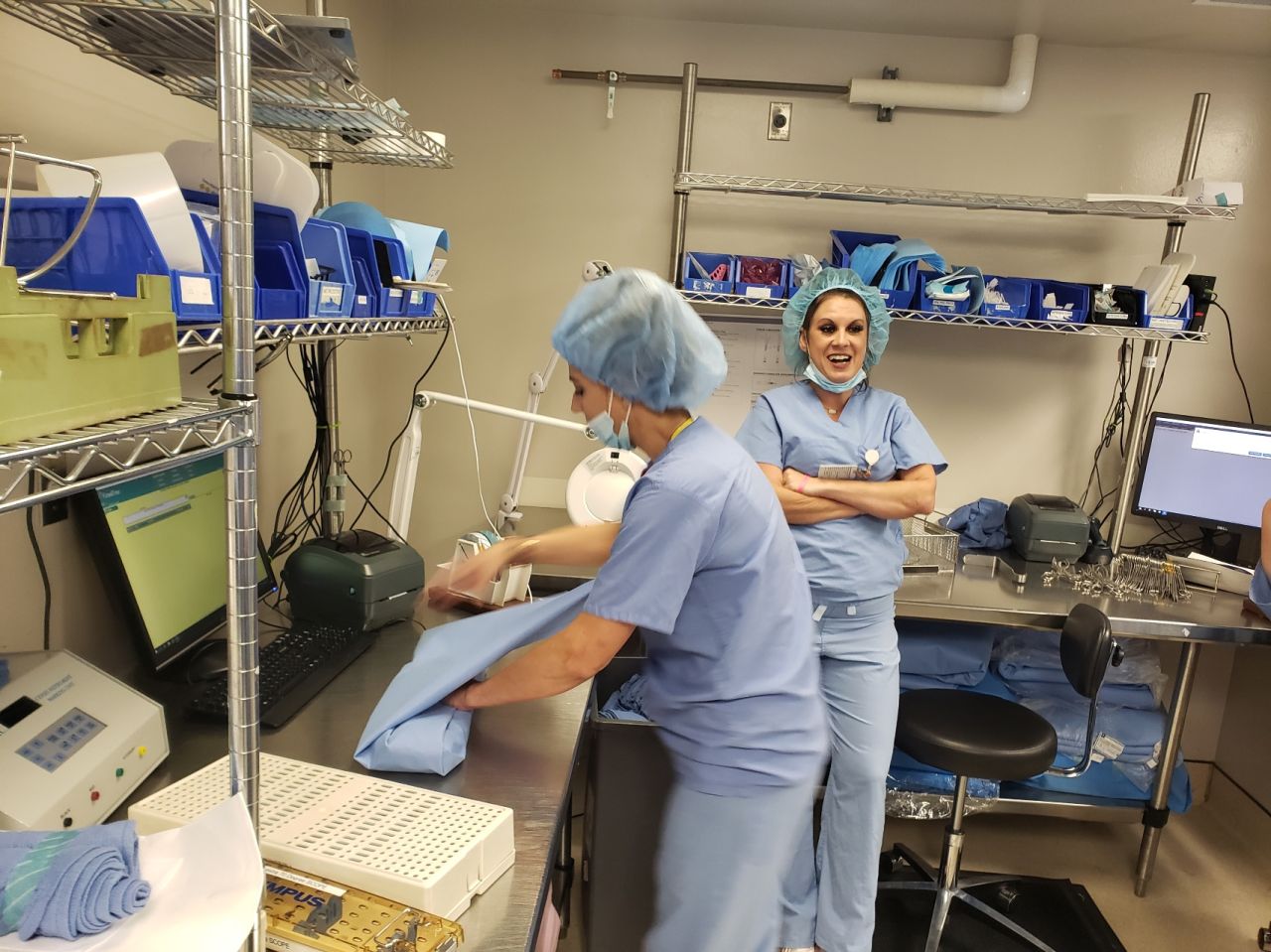
 critical water for ultrasonic rinsing.
critical water for ultrasonic rinsing. et your teammates involved: Next level leadership, infection control, educator (SPD or OR), and a facility Joint Commission readiness team allows your team to holistically engage on behalf of patients, and able to see a 360-degree engaged team view for solving quality problems. Teammates can be of great assistance in training, audits, and keeping SPD updated with regulatory requirements.
et your teammates involved: Next level leadership, infection control, educator (SPD or OR), and a facility Joint Commission readiness team allows your team to holistically engage on behalf of patients, and able to see a 360-degree engaged team view for solving quality problems. Teammates can be of great assistance in training, audits, and keeping SPD updated with regulatory requirements. One set at a time: Take a basic, frequently used set, and verify you have all the reference numbers. Standardize the products in these sets and then move on to the next. Use your primary instrument vendor for data and on-site assistance. Utilize their services for maintaining stock, most-used items, and assisting with pegboard creation and maintenance.
One set at a time: Take a basic, frequently used set, and verify you have all the reference numbers. Standardize the products in these sets and then move on to the next. Use your primary instrument vendor for data and on-site assistance. Utilize their services for maintaining stock, most-used items, and assisting with pegboard creation and maintenance.
 SPD leaders can show industry recommendations to gain additional full time staff support. Time studies regarding the actual safe handling and inspection of instruments with reference to the IFU need to be published to back that need up. Without that information, a question arises: If we could do it with three people before, why do we need six? The fact is, no one wants to admit that processes have not been done correctly, completely, or safely in the past. Quality product was often more a result of luck than best practice or MFC adherence. Most departments don’t have the time, people power, or wherewithal to perform their own holistic time and motion studies to validate. Time and again we return to “what we have been doing is good enough.” Only recently has the “people factor” in the work environment been brought to light on our most important assets, thanks to the AAMI focus on maintaining safe body temperatures, more frequent breaks, and hydration necessities.
SPD leaders can show industry recommendations to gain additional full time staff support. Time studies regarding the actual safe handling and inspection of instruments with reference to the IFU need to be published to back that need up. Without that information, a question arises: If we could do it with three people before, why do we need six? The fact is, no one wants to admit that processes have not been done correctly, completely, or safely in the past. Quality product was often more a result of luck than best practice or MFC adherence. Most departments don’t have the time, people power, or wherewithal to perform their own holistic time and motion studies to validate. Time and again we return to “what we have been doing is good enough.” Only recently has the “people factor” in the work environment been brought to light on our most important assets, thanks to the AAMI focus on maintaining safe body temperatures, more frequent breaks, and hydration necessities.